My heart was racing.
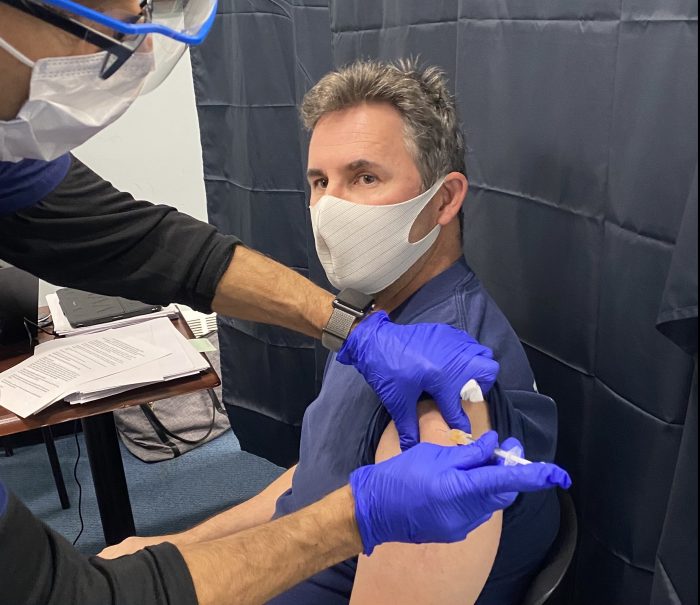
I felt every emotion possible as I took my seat next to the table full of syringes and vials of the COVID-19 vaccine.
The unmistakable odor of sterile alcohol swabs filled the room. The cold steel of hypodermic needles matched the cool uncertainty of my decision.
One week ago, I had decided against getting the vaccine, but now I sit watching the medical assistant draw the just-approved solution from the vial.
My last second doubts would soon be irrelevant as the sharp tip of the needle pierced my skin, and a syringe full of hope emptied into my arm.
As a paramedic on the front lines, I was eligible to be among the first to receive one of the two approved COVID vaccines, a two-shot series produced by both Pfizer and Moderna. I had to decide quickly and experienced bouts of anxiety over the decision. I decided to pass on the vaccine until a later date. The truth is, I was afraid.
How could they develop a safe vaccine in record time?
Was the fast approval a political ploy to try to win an election?
Do we know enough about side-effects?
Do I want to be a test subject for an experimental vaccine when I can wait until they have more research?
I felt uncomfortable with both sides of the decision. The vaccine felt rushed, and I feared they cut corners and skipped steps that might affect safety. But my area is also caught in the throes of another COVID surge, and I’m regularly taking very sick patients to the hospital—and not all of them survive.
While I worry about the safety of the vaccine, I also worry about getting COVID—there are no guarantees, no matter how young or healthy you might be.
I decided to take some time to research everything I could about the vaccines to address my concerns:
How the hell did they develop and approve a vaccine in less than a year without sacrificing safety?
Usually, companies take on huge financial risk when developing new vaccines. The government essentially backstopped this risk by agreeing to purchase the vaccines, whether they proved effective or not. This reduction in financial risk allowed pharmaceutical companies to develop the vaccines more quickly.
The COVID vaccines are a new type that uses Messenger RNA (mRNA) technology, which has been in development for over a decade. This technology is much more efficient than traditional vaccines, allowing them to be produced more rapidly than ever before.
As far as finding test subjects for the studies, it turns out that in the middle of a pandemic, there are plenty of people to enroll in trials and enough of the virus raging through the population to show statistically significant benefits. The trials were not rushed, and approval of the vaccines did not happen until the results of the Phase-3 trials—the ones that prove a clear benefit—were released.
There seem to be reports of serious side effects like life-threatening allergic reactions and Bell’s Palsy.
The CDC reported that some people are at risk for an anaphylactic reaction—a severe, life-threatening allergic reaction that requires an injection of epinephrine to reverse. There is a small percentage of people who are at risk for this reaction.
To combat this, safeguards have been put in place—the vaccine is given in a hospital and monitored for 15-30 minutes—long enough to detect signs of this reaction. If someone does have it, they’re already in the ideal place to treat it.
Bell’s Palsy is a facial nerve inflammation that affects one side of the face and mimics a stroke. It’s a rare condition caused by a viral infection that anyone can get. It’s scary but usually reverses itself with time. During clinical trials, eight incidents of Bell’s Palsy were reported out of almost 50,000 participants—this is on par with the rate seen in the general public, so no link can be established between the vaccine and Bell’s Palsy.
Other side effects are minor. I felt a mild soreness in my arm for two days following the shot. Saying it felt like a punch in the arm is overstating the discomfort. I also felt a bit tired for a couple of days—I didn’t feel sick, but I didn’t feel quite right either. I had no other effects and felt better than I usually do when I get the regular flu vaccine.
Weakness, fever, and muscle aches are also possible side effects, but these effects signal that your immune system is revving up—the vaccine is working.
How effective is the vaccine?
The COVID vaccine is reported to be 95 percent effective—for comparison, the measles vaccine is 97 percent effective, and that was enough to largely wipe out that disease. The COVID vaccine was studied in tens of thousands of people across genders, ages, and races in multiple countries. The Phase-3 studies were randomized, double-blind, and placebo-controlled—the “Gold Standard” for trials.
After I’d scienced the crap out of the vaccine, I felt more comfortable but was still nervous—we’ll know much more than we know now after millions of doses are given and more time has gone by. By then, it’s always possible that more safety concerns will be raised. But in the meantime, we have to weigh the risk of the vaccine versus the risk of getting COVID and spreading it to others.
It’s a personal choice, and no one can make that decision for us.
I understand why someone would decide against the vaccine, and for those who are pregnant, there isn’t much data to assess safety. My decision to get the vaccine didn’t come lightly. For the better part of a year, COVID has turned our lives upside down, and this silver lining comes with uncertainty.
But the truth is, as soon as I was vaccinated, I felt empowered for the first time since this began. I gave a big f*ck you to COVID as I left the hospital, as hope coursed through my veins.
I chose the vaccine to step up my fight against the virus—not just for me, but for my family and friends, my community, our country, and our world.
It was my way of doing everything I can to express love and connection for everyone affected by COVID and helping to rid us of this plague once and for all.
~
Keep yourself up to date with vaccine information here.








Read 53 comments and reply